 
If conversions looks difficult in this specific case due to English units, we can find density in metric units, then convert it: Examples of gas density at several standard conditions P, T. Molar gas constant R need not be used in the calculation. 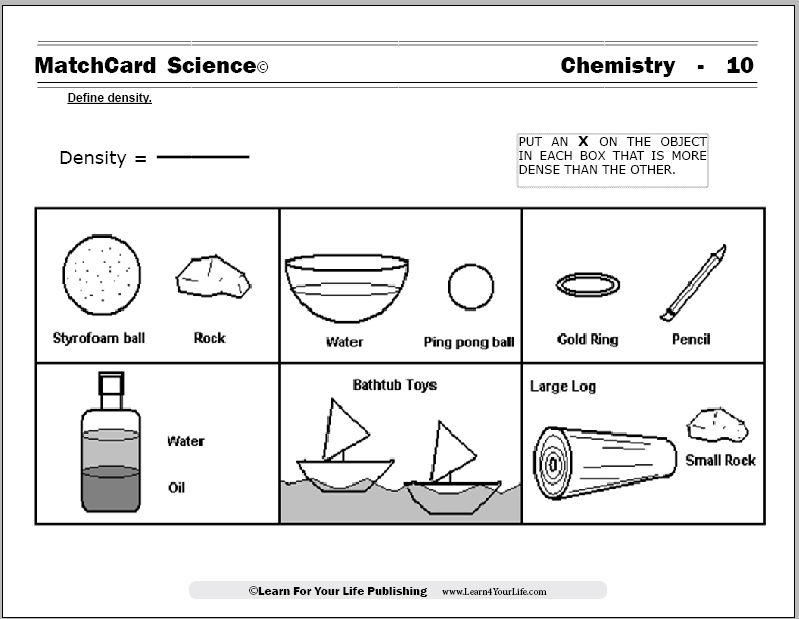
To calculate gas density at some standard condition (or even non standard), we have to find molar volume at this condition by ideal gas law, then divide (α)/(β). A more precise value is 22.414 m3/kgmol, though difference can be insignificant in practical calculations. (α) has mass equal to gas molecular weight MĪs we remember from chemistry, Vm = 22.4 lt/gmol = 22.4 m3/kgmol at 0 oC and 1 Atm a (*). Let us try to make density calculations handy, at least for ideal gas conditions as above, remembering that quantity of 1 kmol of gas Source: Short Cut Formulas For Gas Densities At Various Std.conditions Let me hear comments from some fresh chemical engineers who have just graduated from college. For more details on standard conditions of temperature and pressure please refer the following link: 
It is always good engineering practice to specify the standard conditions in the "Design Basis" document of the particular project being executed. However, this only seems to be applied for natural gas transmission applications in in the United States and Canada. As per the Wikipedia reference provided below, the standard conditions ot temperature utilized by "Organization Of Petroleum Expostring Countries" (OPEC) are 14.73 psia and 60☏. For the upstream oil and gas the most common standard conditions that are utilized are the standard condition 3 when using SI units and standard condition 4 when using English units. The utility of the standard density is to convert standard volume flow in Sm 3 or Scf and volume flow at normal conditions to mass flow by multiplying it with the density, given the standard conditions as mentioned above. Note: The compressibility factor Z is practically unity at all standard conditions mentioned above. 
Note: These are also called normal conditions and the volume defined as Nm 3. (R*T*Z/P) varies and below are a few examples: I will try to summarize these below:Īs a basic we are all supposed to know that gas density is calculated as:įor different standard conditions the denominator in equation 2, i.e. I have been posed by many young entry level engineers about standard gas density calculations which I do in a jiffy using a calculator. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
